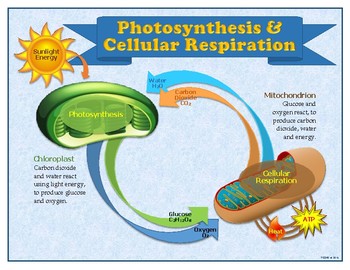
Īerobic metabolism is up to 15 times more efficient than anaerobic metabolism (which yields 2 molecules of ATP per 1 molecule of glucose). However, this maximum yield is never quite reached because of losses due to leaky membranes as well as the cost of moving pyruvate and ADP into the mitochondrial matrix, and current estimates range around 29 to 30 ATP per glucose. Biology textbooks often state that 38 ATP molecules can be made per oxidized glucose molecule during cellular respiration (2 from glycolysis, 2 from the Krebs cycle, and about 34 from the electron transport system). This potential is then used to drive ATP synthase and produce ATP from ADP and a phosphate group. The energy released is used to create a chemiosmotic potential by pumping protons across a membrane. Most of the ATP produced by aerobic cellular respiration is made by oxidative phosphorylation. The potential of NADH and FADH 2 is converted to more ATP through an electron transport chain with oxygen and protons (hydrogen) as the " terminal electron acceptors". The negative ΔG indicates that the reaction is exothermic ( exergonic) and can occur spontaneously. 
Mass balance of the global reaction:Ĭ 6H 12O 6 (s) + 6 O 2 (g) → 6 CO 2 (g) + 6 H 2O (l) + energy The products of this process are carbon dioxide and water, and the energy transferred is used to make bonds between ADP and a third phosphate group to form ATP ( adenosine triphosphate), by substrate-level phosphorylation, NADH and FADH 2. Although carbohydrates, fats and proteins are consumed as reactants, aerobic respiration is the preferred method of pyruvate production in glycolysis, and requires pyruvate to the mitochondria in order to be fully oxidized by the citric acid cycle. The chemical energy stored in ATP (the bond of its third phosphate group to the rest of the molecule can be broken allowing more stable products to form, thereby releasing energy for use by the cell) can then be used to drive processes requiring energy, including biosynthesis, locomotion or transportation of molecules across cell membranes.Īerobic respiration requires oxygen (O 2) in order to create ATP. Nutrients that are commonly used by animal and plant cells in respiration include sugar, amino acids and fatty acids, and the most common oxidizing agent is molecular oxygen (O 2). Although cellular respiration is technically a combustion reaction, it is an unusual one because of the slow, controlled release of energy from the series of reactions. The overall reaction occurs in a series of biochemical steps, some of which are redox reactions. Respiration is one of the key ways a cell releases chemical energy to fuel cellular activity. The reactions involved in respiration are catabolic reactions, which break large molecules into smaller ones, producing large amounts of energy (ATP). Respiration can be either aerobic, requiring O 2 or anaerobic some organisms can switch between aerobic and anaerobic respiration. Ĭellular respiration is a vital process that occurs in the cells of all living organisms. Cellular respiration may be described as a set of metabolic reactions and processes that take place in the cells of organisms to convert chemical energy from nutrients into ATP, and then release waste products.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
